An evening with Jens Juul Holst: GLP-1, women’s health, and the evidence we still need
GLP-1 medicines have changed the way we understand obesity, diabetes and cardio-metabolic risk. But if we view the GLP-1 story through a women’s health lens, it becomes something bigger: a cross-cutting biology with implications for systemic inflammation, cardiovascular disease, fatty liver disease, kidney health, and potentially neuroprotection through anti-inflammatory and metabolic mechanisms currently under investigation in human trials. These are all areas that shape women’s health across the life span.
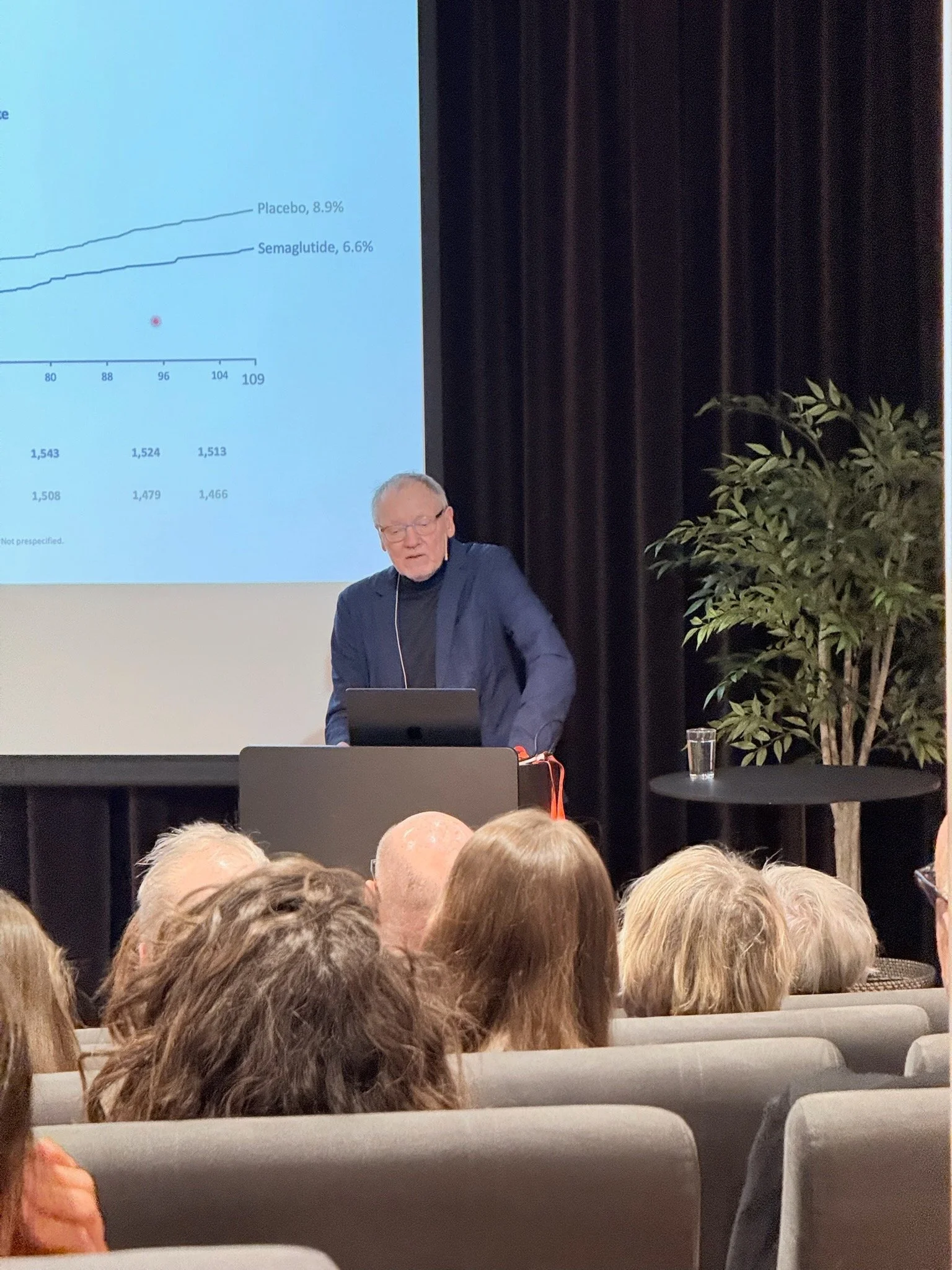
On February 11th, Women in Life Science Norway (WiLD Norway) and The Life Science Cluster hosted a side-event to the 2026 Norway Life Science conferencehosted by Oslo Science City and University of Oslo. It was an evening with Danish Professor Jens Juul Holst, whose scientific discovery of GLP-1 has paved the way for today’s therapies. Holst guided us through biology, research and clinical evidence, and helped to put GLP-1 into a broader framework: obesity is not a single risk factor, but a systemic, multi-organ condition linked to metabolic, mechanical, and mental comorbidities.
Obesity as an overall risk factor
While originally intended for diabetes, a key theme on GLP-1 RAs is that obesity is associated with a long list of comorbidities ranging from cardiovascular disease and type 2 diabetes to renal disease, fatty liver disease, infertility, arthrosis, sleep apnea, anxiety, and depression.
Professor Holst explained a central mechanistic concept called “lipid overflow.” When energy surplus meets limited capacity for safe subcutaneous “pinchable” fat expansion, fat is instead deposited in and immediately surrounding organs. This is called ectopic or “hidden”fat, including visceral abdominal fat that surrounds the abdominal organs, as well as deposited in the liver, heart, pancreas, and even in muscles. Ectopic fat distribution contributes to insulin resistance, dyslipidemia, hypertension, and increased cardiometabolic risk through cellular stress. In other words: the clinical burden is not “extra kilos,” but the systemic impact on multiple organs.
This framing is particularly relevant for women in perimenopause and post-menopause. During these years of hormonal shifts, there is a marked increase in tendency to store ectopic and visceral fat, significantly shifting metabolic and cardiometabolic risk, even if body weight increase is modest. This is one reason why GLP-1 science is thus a women’s health conversation: because cardiometabolic risk shifts significantly during the menopausal transition, GLP-1–based therapies may become particularly relevant in midlife women. Because women spend a larger proportion of life in post-reproductive years, interventions that modify cardiometabolic risk during and after menopause may also have disproportionate long-term public health implications.
How GLP-1 works
The lecture also addressed how appetite is regulated by both homeostatic signals (hormones, glucose, gut signals) and “hedonic drivers” (reward, stress, conditioned cues). It is a misnomer to assume that weight loss on GLP-1 RAs is due directly to slower gut motility and gastric emptying. Instead, GLP-1 reduces food intake through the gut-brain mechanisms, primarily central nervous system pathways. Slides illustrated that GLP-1-based therapies can activate brain regions involved in satiety and reward. As such, not only do individuals experience a loss of appetite, but emerging studies suggest that GLP-1 signaling may modulate pathways involved in substance use behaviors.
“Full body impact”
Holst presented how GLP-1 therapies have moved into outcomes medicine and how large trials show benefits beyond glucose control, including significant reductions in major adverse cardiovascular events in people with type 2 diabetes at high cardiovascular risk, and in individuals with overweight or obesity and established cardiovascular disease. Holst also highlighted progress on kidney outcomes in people with type 2 diabetes and chronic kidney disease, while evidence in non-diabetic kidney disease remains under study.
Several audience questions reflected the real-world excitement for this hot topic, and some of the questions included:
There is increasing pop-science discussion suggesting that gynecologists and endocrinologists are recommending “micro-dosing” or subtherapeutic maintenance dosing of GLP-1 medications for women in perimenopause and menopause, not primarily for weight loss, but for inflammation reduction, sleep improvement, brain fog, fertility, cardiovascular and possible neurovascular protection. Is there evidence to support this practice? Many clinicians see improvements for women, but these effects are not yet supported by formal clinical trials and require systematic investigation.
What are the most common side effects? Most commonly gastrointestinal (such as nausea/vomiting/diarrhea), are often transient, but some rarer events such as gallstones have been reported.
Is GLP-1 for long-term use and will we see a “statin-like” status for these drugs? Where cardiovascular risk is high, the potential benefit may be greatest, but this also raises healthcare system questions about costs, access, prioritization, follow-up and equity. However, oral formulations could reshape access and pricing if scaled effectively.
Will long-term use lead to tolerance and what happens if you stop the treatment? The effect can be maintained while on therapy. Stopping therapy without sustained lifestyle changes is associated with significant weight regain, often with disproportionate fat regain, as opposed to lean mass.
What is the impact on mental health and bone density? Current data do not show an increase in fracture risk, but because weight loss itself can affect bone density, but long-term skeletal effects require continued monitoring.
Could GLP-1 have potential impact on under-explored conditions, e.g. on lipedema? This remains unknown and represents a clear research gap.
Women’s health and inclusion takeaways
WiLD Norway’s “red thread” is that representation affects evidence, and evidence affects who benefits. This event made that link tangible: Gender-specific and life-stage-specific evidence must be standard. Available data suggest modest sex differences in weight loss magnitude and side-effect profiles, but trials have rarely been designed or powered to examine sex-specific cardiovascular or long-term outcomes, despite the widespread knowledge of different presentations for men and women when it comes to cardiometabolic, cardiovascular, and systemic inflammatory diseases. We need trial design, reporting and real-world evidence that take gender and hormonal status/life stage into account, to make sure women, especially in midlife, are not treated as the “averages”. While many patients have been studied and there could be interesting areas to focally disaggregate for women or study women distinctly, this has not yet been widely investigated. However, we know that studies in men have reported improvements in erectile function. Future trials should routinely stratify by sex, menopausal status, hormone therapy use, and reproductive history, variables that are rarely analyzed but are biologically relevant.
Treatment persistence is a challenge in most therapies, but it is not evenly distributed. Real-world data suggest that a substantial proportion of patients discontinue therapy within the first year. Adherence is affected by several practical and behavioral factors such as high out-of-pocket costs, side effects that some find difficult to tolerate, discontinuation once a target weight is reached and, for some, a reduced sense of “food reward.” When access depends on ability to pay, or on whether patients have structured follow-up and support or not, these factors can widen existing health inequities.
At the same time, the period of weight loss represents a critical window for long-term health. Professor Holst’s most practical message was clear: use the treatment phase to establish sustainable habits by improving diet quality, building routines and increasing physical activity, because lasting outcomes require more than medication alone.
Even though long-term pharmacotherapy combined with intensive behavioral support is emphasized in international guidance, medication alone will not solve obesity at a population level either. Thar will require equity and policy choices to shape impact, and place responsibility on systems to ensure fair access, strengthen follow-up and to keep prevention at the core.
Next move for the GLP-1 field?
The GLP-1 field is moving fast. WiLD Norway’s contribution is the same as for all of life science and leadership: to keep asking the questions that ensure progress is inclusive:
Who is represented in studies?
Whose outcomes are measured?
Who gets access?
And who sits at the tables where research priorities, clinical guidelines, and innovation investments are decided?
Thank you to Professor Jens Juul Holst, The Life Science Cluster, University of Oslo, and to everyone who participated with thoughtful questions and perspectives.